Vitamin D - Season-stratified GWAS
# Libraries
library(ggplot2)
library(dplyr)
library(ggrepel)
library(DT)
library(qqman)
library(gridExtra)We hypothesised that if, for example, there were pathways involved in metabolising vitamin D (which would be active in Summer months, when vitamin D production is increased), or in storing vitamin D (which would be active in Winter months, when vitamin D production is limited), there would be different genetic variants associated with vitamin D levels in different seasons. To test that hypothesis, we stratified the UK Biobank data into two cohorts: (1) individuals assessed in Autumn/Winter, and (2) individuals assessed Spring/Summer. Then, we ran fastGWA separately in the two sub-samples. Results are presented below.
Methods
Data
The following files were used to run the analyses:
- Phenotype file: File with one line per individual
and a column with the phenotype. The phenotype was obtained after (1)
restricting to individuals of European ancestry, (2) restricting to
individuals with vitamin D levels measured in the season of interest,
and (3) applying a rank-based inverse-normal transformation (RINT).
Specifically, two phenotype files were used to run two GWAS (more
information on Pheno page):
- “Winter” phenotype - 162591 individuals of European
ancestry
- “Summer” phenotype - 177082 individuals of European
ancestry
- “Winter” phenotype - 162591 individuals of European
ancestry
- Genotype file: We restricted the analyses to
individuals of European ancestry and included 44741804 autossomal
variants + 260713 variants in chromosome X (including the
pseudo-autosomal region). Variants had (1) MAC > 5, (2) INFO >
0.3, and (3) genotype missingness > 0.05. In addition, variants with
missingness < 0.05, pHWE < 1x10-5, and MAC > 5 in
the subset of unrelated European individuals were excluded.
- Sparse GRM: For this analysis we used a sparse GRM calculated using HapMap3 SNPs from individuals of European ancestry.
- Covariates files: This file includes one column per
covariate to fit in the model. Covariates included were: age, sex,
genotyping batch, UKB assessment centre, month of assessment,
information on whether vitamin supplements were ever taken, and the
first 40 PCs. For more detail see the
Covariatessection of the Pheno page.
GWAS
We conducted two GWA analyses with Fast-GWA, restricting to SNPs with MAF>0.01.
# ------ Make mbfile with all individual-level data files to include in the analysis
tmpDir=$WD/results/stratifiedGWAS/tmpDir
genoDir=$medici/UKBiobank/v3EUR_impQC
echo $genoDir/ukbEUR_imp_chr1_v3_impQC > $tmpDir/UKB_v3EUR_impQC.files
for i in `seq 2 22`; do echo $genoDir/ukbEUR_imp_chr${i}_v3_impQC >> $tmpDir/UKB_v3EUR_impQC.files; done
# #########################################
# Run stratified GWAS - autosomes
# #########################################
# Set directories
results=$WD/results/stratifiedGWAS
tmpDir=$results/tmpDir
for season in winter summer
do
# Files/settings to use
prefix=vitD_${season}GWAS
geno=$tmpDir/UKB_v3EUR_impQC.files
pheno=$WD/input/mlm_${season}_pheno
grm=$WD/input/UKB_HM3_m01_sp
quantCovars=$WD/input/quantitativeCovariates
catgCovars=$WD/input/categoricalCovariates
snps2include=$WD/input/UKB_v3EUR_impQC/UKB_EUR_impQC_maf0.01.snplist
output=$results/$prefix
# Run GWAS
cd $results
gcta=$bin/gcta/gcta_1.92.3beta3
tmp_command="$gcta --mbfile $geno \
--fastGWA-lmm \
--pheno $pheno \
--grm-sparse $grm \
--qcovar $quantCovars\
--covar $catgCovars \
--covar-maxlevel 110 \
--extract $snps2include \
--threads 16 \
--out $output"
qsubshcom "$tmp_command" 16 10G $prefix 24:00:00 ""
echo $prefix
done
# Format results
for season in winter summer
do
# Merge results from each chr after all have finished running
prefix=vitD_${season}GWAS
gzip $results/$prefix.fastGWA
mv $results/$prefix.fastGWA.gz $results/$prefix.gz
# Convert GWAS sumstats to .ma format (for COJO)
echo "SNP A1 A2 freq b se p n" > $results/$prefix.ma
zcat $results/$prefix.gz | awk '{print $2,$4,$5,$7,$8,$9,$10,$6}' | sed 1d >> $results/$prefix.ma
done
# #########################################
# Run stratified GWAS - X chromosome
# #########################################
# Set directories
results=$WD/results/stratifiedGWAS
tmpDir=$results/tmpDir
for season in winter summer
do
# Files/settings to use
pheno=$WD/input/mlm_${season}_pheno
grm=$WD/input/UKB_HM3_m01_sp
quantCovars=$WD/input/quantitativeCovariates
catgCovars=$WD/input/categoricalCovariates_noSex
# ---- Run GWAS separately for males and females
cd $results
for sex in FEMALE MALE
do
# Run X chromosome pseudo-autosomal region (chr XY) as well
for region in X XY
do
prefix=vitD_${season}_chr$region.$sex
geno=$medici/v3EUR_chrX/ukbEURv3_${region}_maf0.1_$sex
output=$tmpDir/$prefix
gcta=$bin/gcta/gcta_1.92.3beta3
tmp_command="$gcta --bfile $geno \
--fastGWA-lmm \
--pheno $pheno \
--grm-sparse $grm \
--qcovar $quantCovars\
--covar $catgCovars \
--covar-maxlevel 110 \
--threads 16 \
--out $output"
qsubshcom "$tmp_command" 16 30G $prefix 6:00:00 ""
done # end loop through chr
done # end loop through sex
done # end loop through season
# #########################################
# Meta-analyse results from males and females for X and XY
# #########################################
results=$WD/results/stratifiedGWAS
tmpDir=$results/tmpDir
metal=$bin/metal/metal
# Run meta-analysis
cd $results
for season in winter summer
do
for chr in X XY
do
sumstats1=$tmpDir/vitD_${season}_chr$chr.FEMALE.fastGWA
sumstats2=$tmpDir/vitD_${season}_chr$chr.MALE.fastGWA
output=$tmpDir/vitD_${season}_chr$chr
METALscript=$tmpDir/$season.chr$chr.SEXmeta.METALscript
logFile=$results/job_reports/$season.chr$chr.SEXmeta.log
echo "
CLEAR
SCHEME STDERR
AVERAGEFREQ ON
# Describe and process results for X chromosome
MARKER SNP
ALLELE A1 A2
EFFECT BETA
PVALUE P
WEIGHT N
STDERR SE
FREQLABEL AF1
PROCESS $sumstats1
PROCESS $sumstats2
# Run meta-analysis
OUTFILE $output .TBL
ANALYZE
" > $METALscript
# Run meta-analysis
$metal < $METALscript > $logFile
done # end loop through chromosomes
done # end loop through seasons
# #########################################
# Format meta-analysed results and merge with autossomes
# Generate .ma format files for COJO
# #########################################
cd $WD/results/stratifiedGWAS
R
for(season in c("winter","summer"))
{
prefix=paste0("vitD_",season)
# Read X meta-analyses results + GWAS results for autosomes
xmeta=read.table(paste0("tmpDir/",prefix,"_chrX1.TBL"),h=T,stringsAsFactors=F)
xymeta=read.table(paste0("tmpDir/",prefix,"_chrXY1.TBL"),h=T,stringsAsFactors=F)
autosomes=read.table(paste0(prefix,"GWAS.gz"), h=T,stringsAsFactors=F)
names(xmeta)=c("SNP","A1","A2","AF1","AF1_SE","BETA","SE","P","direction")
names(xymeta)=c("SNP","A1","A2","AF1","AF1_SE","BETA","SE","P","direction")
# Get chrX details (BP and N) from female and male GWAS
xfemale=read.table(paste0("tmpDir/",prefix,"_chrX.FEMALE.fastGWA"), h=T,stringsAsFactors=F)
xmale=read.table(paste0("tmpDir/",prefix,"_chrX.MALE.fastGWA"), h=T,stringsAsFactors=F)
## Get all results in the same order
xmeta=xmeta[match(xfemale$SNP,xmeta$SNP),]
xmale=xmale[match(xfemale$SNP,xmale$SNP),]
## Add BP
xmeta$POS=xfemale$POS
# Add N
xmeta$N=xfemale$N+xmale$N
# Change alleles to capital letters
xmeta$A1=toupper(xmeta$A1)
xmeta$A2=toupper(xmeta$A2)
# Define chromosome
xmeta$CHR=23
# Get chrXY details (BP, and N) from female and male GWAS
xyfemale=read.table(paste0("tmpDir/",prefix,"_chrXY.FEMALE.fastGWA"), h=T,stringsAsFactors=F)
xymale=read.table(paste0("tmpDir/",prefix,"_chrXY.MALE.fastGWA"), h=T,stringsAsFactors=F)
## Get all results in the same order
xymeta=xymeta[match(xyfemale$SNP,xymeta$SNP),]
xymale=xymale[match(xyfemale$SNP,xymale$SNP),]
## Add BP
xymeta$POS=xyfemale$POS
## Add N
xymeta$N=xyfemale$N+xymale$N
# Define chromosome
xymeta$CHR=25
# Change alleles to capital letters
xymeta$A1=toupper(xymeta$A1)
xymeta$A2=toupper(xymeta$A2)
# Make table in fastGWA format
# CHR SNP POS A1 A2 N AF1 BETA SE P
xmeta_fastgwa=xmeta[,c("CHR","SNP","POS","A1","A2","N","AF1","BETA","SE","P")]
xymeta_fastgwa=xymeta[,c("CHR","SNP","POS","A1","A2","N","AF1","BETA","SE","P")]
## Merge X + XY + autosomes
allChr=rbind(autosomes, xmeta_fastgwa, xymeta_fastgwa)
# Make table in COJO (.ma) format
# SNP A1 A2 freq b se p n
## CHR X
xmeta_ma=xmeta[,c("SNP","A1","A2","AF1","BETA","SE","P","N")]
names(xmeta_ma)=c("SNP","A1","A2","freq","b","se","p","n")
## CHR XY
xymeta_ma=xymeta[,c("SNP","A1","A2","AF1","BETA","SE","P","N")]
names(xymeta_ma)=c("SNP","A1","A2","freq","b","se","p","n")
# Save new file formats
## All chromosomes
write.table(allChr, file=gzfile(paste0(prefix,"_withX.gz")), quote=F, row.names=F)
## X and XY in fastGWA format (for clumping)
write.table(xmeta_fastgwa, file=gzfile(paste0("tmpDir/",prefix,"_chrX.gz")), quote=F, row.names=F)
write.table(xymeta_fastgwa, file=gzfile(paste0("tmpDir/",prefix,"_chrXY.gz")), quote=F, row.names=F)
## X and XY in .ma format (for COJO)
write.table(xmeta_ma, paste0(prefix,"_chrX.ma"), quote=F, row.names=F)
write.table(xymeta_ma, paste0(prefix,"_chrXY.ma"), quote=F, row.names=F)
}
COJO
The following options were used in this analysis:
- –bfile: We used the GWAS individual-level genotype data as reference.
- –cojo-slct: Performs a step-wise model selection of independently associated SNPs
- –cojo-p 5e-8 (default): P-value to declare a genome-wide significant hit
- –cojo-wind 10,000 (default): Distance (in KB) from which SNPs are considered in linkage equilibrium
- –cojo-collinear 0.9 (default): maximum r2 to be considered an independent SNP, i.e. SNPs with r2>0.9 will not be considered further
- –diff-freq 0.2 (default): maximum allele difference bet
- extract: Restrict analysis to SNPs with MAF>0.01
# #########################################
# Run COJO on GWAS results
# #########################################
# Directories
results=$WD/results/stratifiedGWAS
tmpDir=$results/tmpDir
# Files/settings to use
random20k_dir=$WD/input/UKB_v3EURu_impQC/random20K
ldRef=$random20k_dir/ukbEURu_imp_chr{TASK_ID}_v3_impQC_random20k_maf0.01
filters=maf0.01
snps2include=$WD/input/UKB_v3EUR_impQC/UKB_EUR_impQC_$filters.snplist
for season in summer winter
do
prefix=vitD_${season}GWAS
inFile=$results/$prefix.ma
outFile=$tmpDir/${prefix}_${filters}_chr{TASK_ID}
# Run COJO
cd $results
gcta=$bin/gcta/gcta_1.92.3beta3
tmp_command="$gcta --bfile $ldRef \
--cojo-slct \
--cojo-file $inFile \
--extract $snps2include \
--out ${outFile}"
qsubshcom "$tmp_command" 1 100G ${prefix}_COJO 24:00:00 "-array=1-22"
echo ${prefix}_COJO
done
# Format results
for season in summer winter
do
prefix=vitD_${season}GWAS
# Merge COJO results once all chr finished running
cat $tmpDir/${prefix}_${filters}*.jma.cojo | grep "Chr" | uniq > $results/${prefix}_${filters}.cojo
cat $tmpDir/${prefix}_${filters}_chr*.jma.cojo | grep -v "Chr" |sed '/^$/d' >> $results/${prefix}_${filters}.cojo
done
LDSC
We ran bivariate LDSC regression with both GWAS results to assess the genetic correlation between the two.
# ##############################
# Run bivariate LDSC regression
# ##############################
module load ldsc
# Directories
results=$WD/results/stratifiedGWAS
tmpDir=$results/tmpDir
ldscDir=$bin/ldsc
cd $results
for season in summer winter
do
# Format GWAS results
prefix=vitD_${season}GWAS
echo "SNP A1 A2 b n p" > $tmpDir/$prefix.ldHubFormat
awk 'NR>1 {print $1,$2,$3,$5,$8,$7}' $results/$prefix.ma >> $tmpDir/$prefix.ldHubFormat
# Munge GWAS sumstats (process sumstats and restrict to HapMap3 SNPs)
munge_sumstats.py --sumstats $tmpDir/$prefix.ldHubFormat \
--merge-alleles $ldscDir/w_hm3.snplist \
--out $tmpDir/$prefix
done
# Run rg
ldsc.py --rg $tmpDir/vitD_winterGWAS.sumstats.gz,$tmpDir/vitD_summerGWAS.sumstats.gz \
--ref-ld-chr $ldscDir/eur_w_ld_chr/ \
--w-ld-chr $ldscDir/eur_w_ld_chr/ \
--out $results/vitD_stratifiedGWAS.ldsc.rg
In addition, we ran LDSC on each GWAS (i.e. ‘Summer’ and ‘Winter’, separately), to determine proportion of phenotypic variance they explained. To run LDSC, we restricted the GWAS summary statistics to variants found in w_hm3.noMHC.snplist (downloaded from LDHub). This is a list of HapMap3 SNPs and excludes SNPs in the MHC region.
module load ldsc
# Directories
results=$WD/results/stratifiedGWAS
tmpDir=$results/tmpDir
ldscDir=$bin/ldsc
cd $results
for season in summer winter
do
# Format GWAS results
prefix=vitD_${season}GWAS
awk '{print $1,$2,$3,$5,$8,$7}' $results/$prefix.ma > $tmpDir/$prefix.ldHubFormat
# Restrict to common SNPs
sed 1q $tmpDir/$prefix.ldHubFormat > $results/$prefix.ldHubFormat
awk 'NR==FNR{a[$1];next} ($1 in a)' $WD/input/UKB_v3EURu_impQC/UKB_EURu_impQC_maf0.01.snplist <(awk 'NR==FNR{a[$1];next} ($1 in a)' $WD/input/w_hm3.noMHC.snplist $tmpDir/$prefix.ldHubFormat) >> $results/$prefix.ldHubFormat
# Munge sumstats (process sumstats and restrict to HapMap3 SNPs)
munge_sumstats.py --sumstats $results/$prefix.ldHubFormat \
--merge-alleles $ldscDir/w_hm3.snplist \
--out $tmpDir/$prefix
# Run LDSC
ldsc.py --h2 $tmpDir/$prefix.sumstats.gz \
--ref-ld-chr $ldscDir/eur_w_ld_chr/ \
--w-ld-chr $ldscDir/eur_w_ld_chr/ \
--out $results/$prefix.ldsc
done
Results
Note: All results presented are for variants with MAF>0.01.
Genetic correlation
# Read-in bivariate LDSC regression results
tmp=read.table("results/stratifiedGWAS/vitD_stratifiedGWAS.ldsc.rg.log", skip=30, fill=T, h=F, colClasses="character", sep="_")[,1]
# Get results:
intercept=strsplit(grep("Intercept",tmp, value=T)[3],":")[[1]][2]
rg=strsplit(grep("Genetic Correlation:",tmp, value=T),":")[[1]][2]We ran bivariate LDSC regression with both GWAS results to assess the genetic correlation between the two:
- Bivariate LDSC Intercept: 0.011 (0.0189)
- Genetic Correlation: 0.8941 (0.0517)
The genetic correlation between the two traits is high, indicating
that the genetic background associated with vitamin D levels in winter
is similar to the genetic background associated with vitamin D levels in
summer.
Manhattan plot
Summer GWAS
# Read in GWAS results ---------------------------
#gwas=read.table("results/stratifiedGWAS/vitD_summer_withX.gz", h=T, stringsAsFactors=F)
#saveRDS(gwas, file="results/stratifiedGWAS/vitD_summer_withX.RDS")
gwas=readRDS("results/stratifiedGWAS/vitD_summer_withX.RDS")
names(gwas)[grep("POS",names(gwas))]="BP"
cojo=read.table("results/stratifiedGWAS/vitD_summerGWAS_maf0.01.cojo", h=T, stringsAsFactors=F)
# Add cumulative position + clumping/cojo information for plotting
# ----- Add chromosome start site to the GWAS result
don=aggregate(BP~CHR,gwas,max)
don$tot=cumsum(as.numeric(don$BP))-don$BP
don=merge(gwas,don[,c("CHR","tot")])
# ----- Add a cumulative position to plot in this order
don=don[order(don$CHR,don$BP),]
# ----- Restrict SNPs with P<0.03 + a random samples of 100K SNPs (to plot faster)
don=don[don$P<0.03 | don$SNP %in% sample(gwas$SNP,100000),]
don$BPcum=don$BP+don$tot
# ----- Add cojo information
don$is_cojo_sig=ifelse(don$SNP %in% cojo$SNP & don$P<1e-15, "yes","no")
don$is_cojo_nonSig=ifelse(don$SNP %in% cojo$SNP & don$P>5e-8, "yes","no")
don$is_cojo=ifelse(don$SNP %in% cojo$SNP, "yes","no")
# ----- Prepare X axis
axisdf = don %>% group_by(CHR) %>% summarize(center=( max(BPcum) + min(BPcum) ) / 2 )
# Generate Manhattan plot
ggplot(don, aes(x=BPcum, y=-log10(P))) +
geom_point( aes(color=as.factor(CHR)), alpha=0.3, size=0.5) +
scale_color_manual(values = rep(c("lightsteelblue4", "lightsteelblue3"),44)) +
# Highlighted SNPs of interest
geom_point(data=don[don$is_cojo=="yes",], color="red", size=2) +
geom_label_repel( data=don[don$is_cojo_sig=="yes",], aes(label=SNP), size=2) +
# Make X axis
scale_x_continuous( label=axisdf$CHR, breaks=axisdf$center ) +
labs(x="")+
theme_bw() +
theme(legend.position="none",
panel.border = element_blank(),
panel.grid.major.x = element_blank(),
panel.grid.minor.x = element_blank())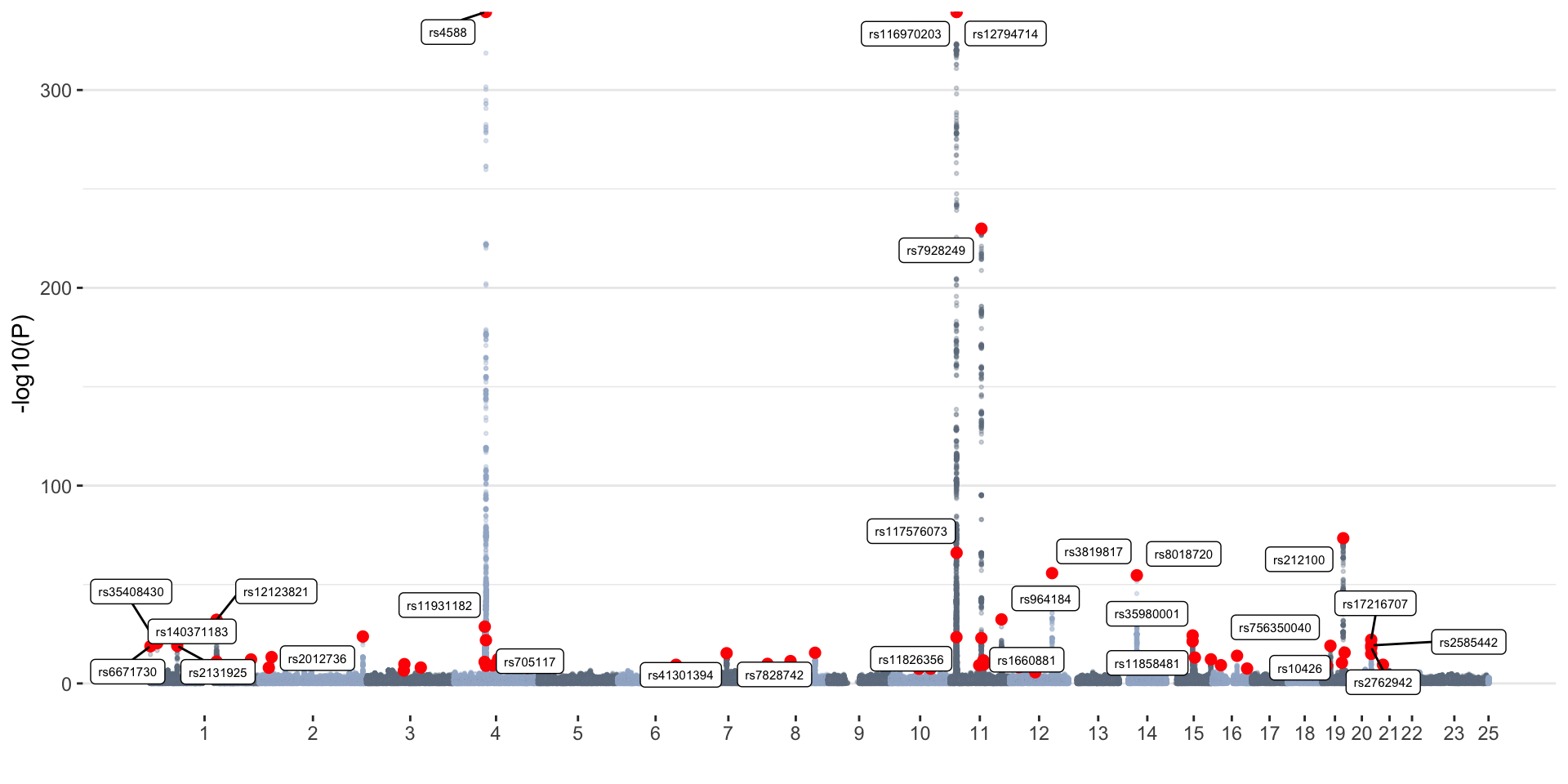
# Keep info to summarise in the report below
summer_common=gwas
summer_cojo_common=cojoRed dots represent independent associations
identified with COJO. Top independent SNPs (P<1x10-15) are
labeled.
Winter GWAS
# Read in GWAS results ---------------------------
#gwas=read.table("results/stratifiedGWAS/vitD_winter_withX.gz", h=T, stringsAsFactors=F)
#saveRDS(gwas, "results/stratifiedGWAS/vitD_winter_withX.RDS")
gwas=readRDS("results/stratifiedGWAS/vitD_winter_withX.RDS")
names(gwas)[grep("POS",names(gwas))]="BP"
cojo=read.table("results/stratifiedGWAS/vitD_winterGWAS_maf0.01.cojo", h=T, stringsAsFactors=F)
# Add cumulative position + clumping/cojo information for plotting
# ----- Add chromosome start site to the GWAS result
don=aggregate(BP~CHR,gwas,max)
don$tot=cumsum(as.numeric(don$BP))-don$BP
don=merge(gwas,don[,c("CHR","tot")])
# ----- Add a cumulative position to plot in this order
don=don[order(don$CHR,don$BP),]
# ----- Restrict SNPs with P<0.03 + a random samples of 100K SNPs (to plot faster)
don=don[don$P<0.03 | don$SNP %in% sample(gwas$SNP,100000),]
don$BPcum=don$BP+don$tot
# ----- Add cojo information
don$is_cojo_sig=ifelse(don$SNP %in% cojo$SNP & don$P<1e-15, "yes","no")
don$is_cojo_nonSig=ifelse(don$SNP %in% cojo$SNP & don$P>5e-8, "yes","no")
don$is_cojo=ifelse(don$SNP %in% cojo$SNP, "yes","no")
# ----- Prepare X axis
axisdf = don %>% group_by(CHR) %>% summarize(center=( max(BPcum) + min(BPcum) ) / 2 )
# Generate Manhattan plot
ggplot(don, aes(x=BPcum, y=-log10(P))) +
geom_point( aes(color=as.factor(CHR)), alpha=0.3, size=0.5) +
scale_color_manual(values = rep(c("lightsteelblue4", "lightsteelblue3"),44)) +
# Highlighted SNPs of interest
geom_point(data=don[don$is_cojo=="yes",], color="red", size=2) +
geom_label_repel( data=don[don$is_cojo_sig=="yes",], aes(label=SNP), size=2) +
# Make X axis
scale_x_continuous( label=axisdf$CHR, breaks=axisdf$center ) +
labs(x="")+
theme_bw() +
theme(legend.position="none",
panel.border = element_blank(),
panel.grid.major.x = element_blank(),
panel.grid.minor.x = element_blank())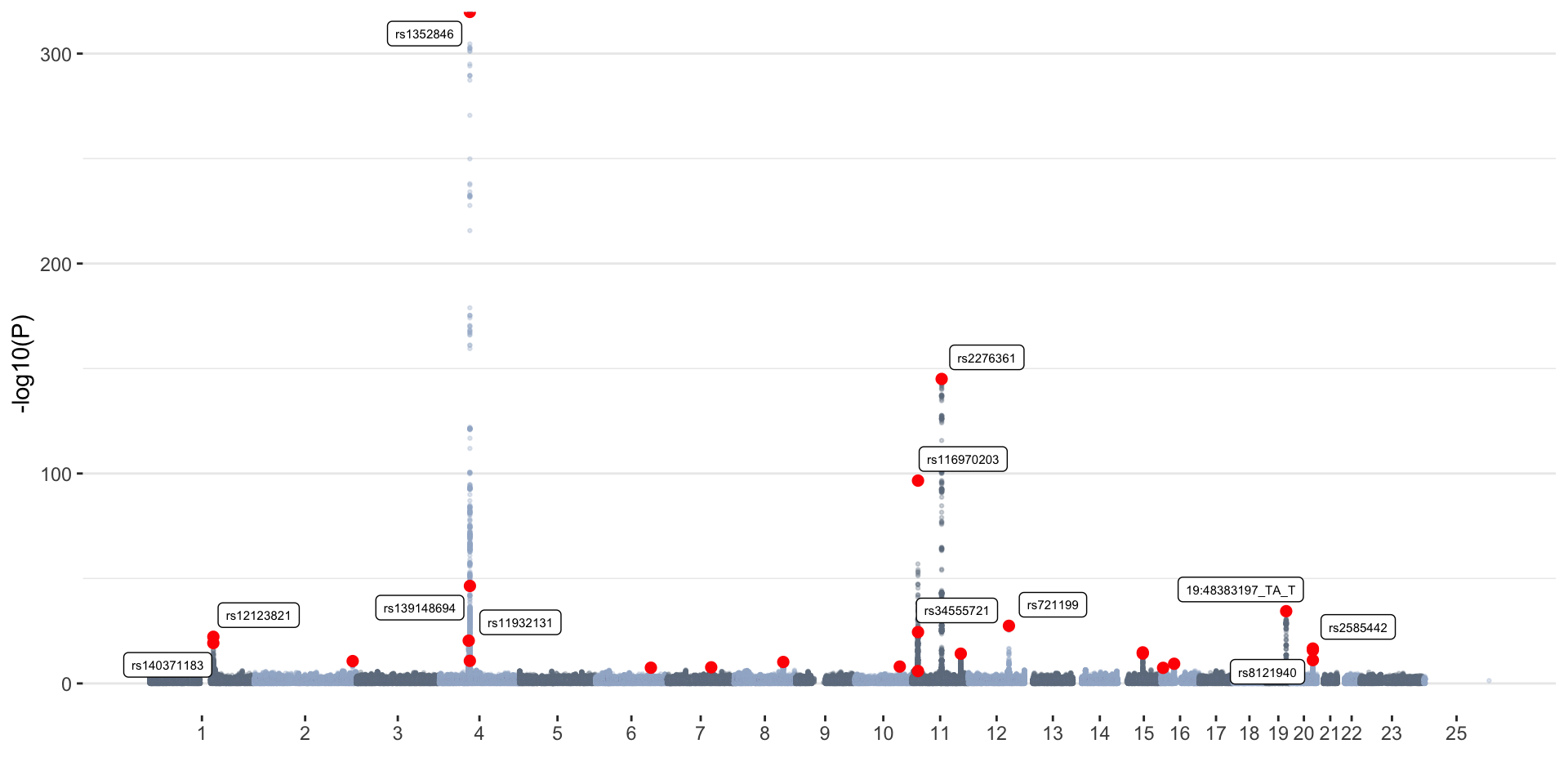
# Keep info to summarise in the report below
winter_common=gwas
winter_cojo_common=cojoRed dots represent independent associations
identified with COJO. Top independent SNPs (P<1x10-15) are
labeled.
Summary of results
Summer GWAS
- 8806780 variants tested
- 12331 GWS (P<5x10-8) hits
- 70 independent associations (determined with COJO)
- 65 independent associations (determined with COJO) that were GWS in the original GWAS
indep = summer_cojo_common %>% arrange(Chr, bp)
# Format table
indep=indep[,c("Chr","SNP","bp","refA","freq","b","se","p","n","freq_geno","bJ","bJ_se","pJ")]
indep$p=format(indep$p, digits=3)
indep$pJ=format(indep$pJ, digits=3)
indep=indep %>% mutate_at(vars(se, bJ_se, b, bJ), round, 4)
indep=indep %>% mutate_at(vars(freq, freq_geno), round, 2)
# Present table
datatable(indep,
rownames=FALSE,
extensions=c('Buttons','FixedColumns'),
options=list(pageLength=5,
dom='frtipB',
buttons=c('csv', 'excel'),
scrollX=TRUE),
caption="List of independent associations in summer GWAS") %>%
formatStyle("p","white-space"="nowrap") %>%
formatStyle("pJ","white-space"="nowrap") Columns are: Chr, chromosome; SNP, SNP rs ID; bp, physical position; refA, the effect allele; type, type of QTL; freq, frequency of the effect allele in the original data; b, se and p, effect size, standard error and p-value from the original GWAS; n, estimated effective sample size; freq_geno, frequency of the effect allele in the reference sample; bJ, bJ_se and pJ, effect size, standard error and p-value from a joint analysis of all the selected SNPs.
Winter GWAS
- 8806780 variants tested
- 3993 GWS (P<5x10-8) hits
- 26 independent associations (determined with COJO)
- 25 independent associations (determined with COJO) that were GWS in the original GWAS
indep = winter_cojo_common %>% arrange(Chr, bp)
# Format table
indep=indep[,c("Chr","SNP","bp","refA","freq","b","se","p","n","freq_geno","bJ","bJ_se","pJ")]
indep$p=format(indep$p, digits=3)
indep$pJ=format(indep$pJ, digits=3)
indep=indep %>% mutate_at(vars(se, bJ_se, b, bJ), round, 4)
indep=indep %>% mutate_at(vars(freq, freq_geno), round, 2)
# Present table
datatable(indep,
rownames=FALSE,
extensions=c('Buttons','FixedColumns'),
options=list(pageLength=5,
dom='frtipB',
buttons=c('csv', 'excel'),
scrollX=TRUE),
caption="List of independent associations in winter GWAS") %>%
formatStyle("p","white-space"="nowrap") %>%
formatStyle("pJ","white-space"="nowrap") Columns are: Chr, chromosome; SNP, SNP rs ID; bp, physical position; refA, the effect allele; type, type of QTL; freq, frequency of the effect allele in the original data; b, se and p, effect size, standard error and p-value from the original GWAS; n, estimated effective sample size; freq_geno, frequency of the effect allele in the reference sample; bJ, bJ_se and pJ, effect size, standard error and p-value from a joint analysis of all the selected SNPs.
QQplot + LDSC
Summer GWAS
gwas=summer_common
names(gwas)[grep("p",names(gwas))]="P"
pthrh=0.03
tmp=c(gwas[gwas$P<pthrh,"P"], sample(gwas[gwas$P>pthrh,"P"],100000))
qq(tmp)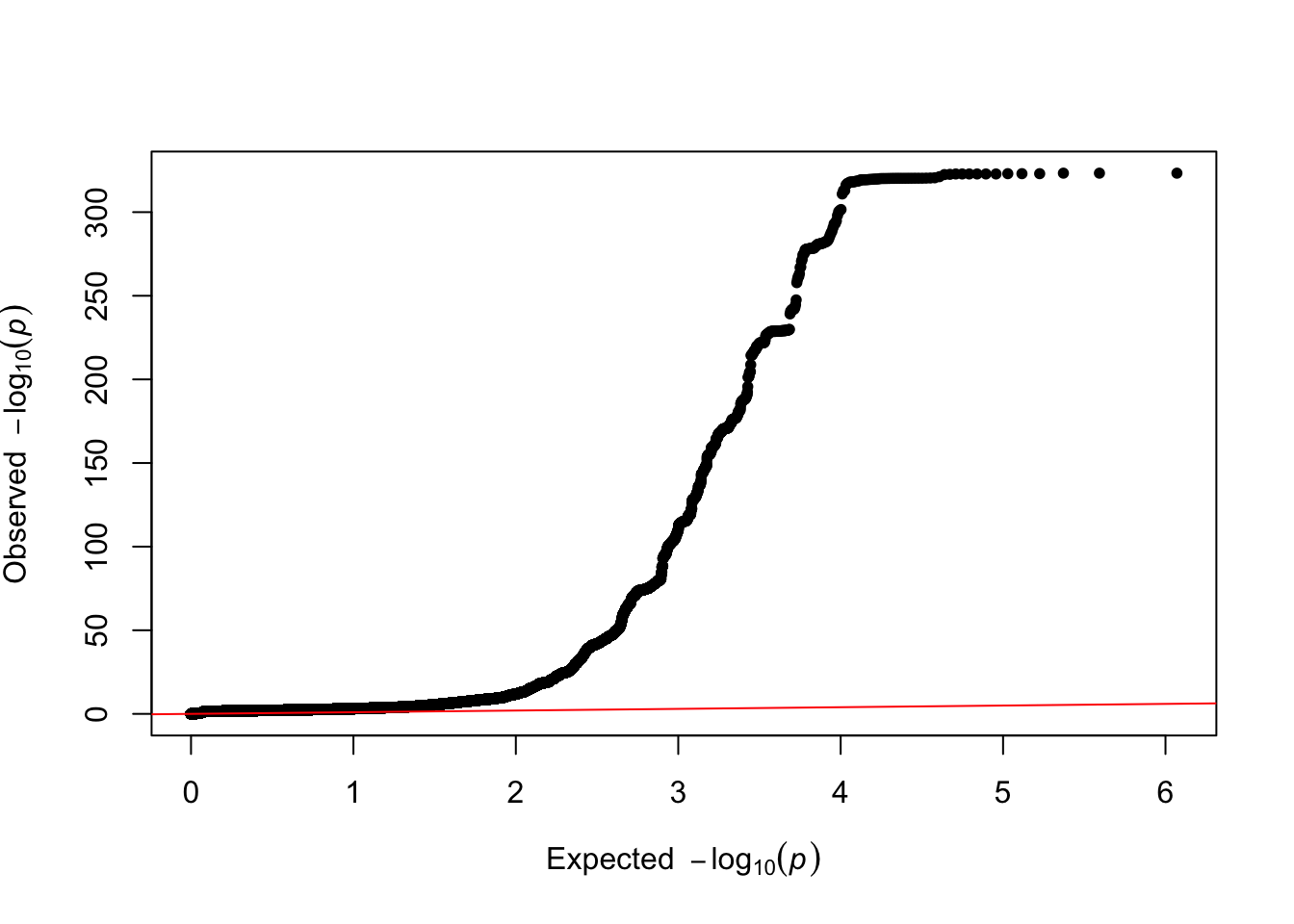
LDSC
tmp=read.table("results/stratifiedGWAS/vitD_summerGWAS.ldsc.log", skip=15, fill=T, h=F, colClasses="character", sep="_")[,1]
# Get results:
h2=strsplit(grep("Total Observed scale h2",tmp, value=T),":")[[1]][2]
lambda_gc=strsplit(grep("Lambda GC",tmp, value=T),":")[[1]][2]
chi2=strsplit(grep("Mean Chi",tmp, value=T),":")[[1]][2]
intercept=strsplit(grep("Intercept",tmp, value=T),":")[[1]][2]
ratio=strsplit(grep("Ratio",tmp, value=T),":")[[1]][2]- Total Observed scale h2: 0.1389 (0.0293)
- Lambda GC: 1.2564
- Mean Chi2: 1.5253
- Intercept: 1.0263 (0.0119)
- Ratio: 0.05 (0.0226)
Winter GWAS
gwas=winter_common
names(gwas)[grep("p",names(gwas))]="P"
pthrh=0.03
tmp=c(gwas[gwas$P<pthrh,"P"], sample(gwas[gwas$P>pthrh,"P"],100000))
pw=qq(tmp)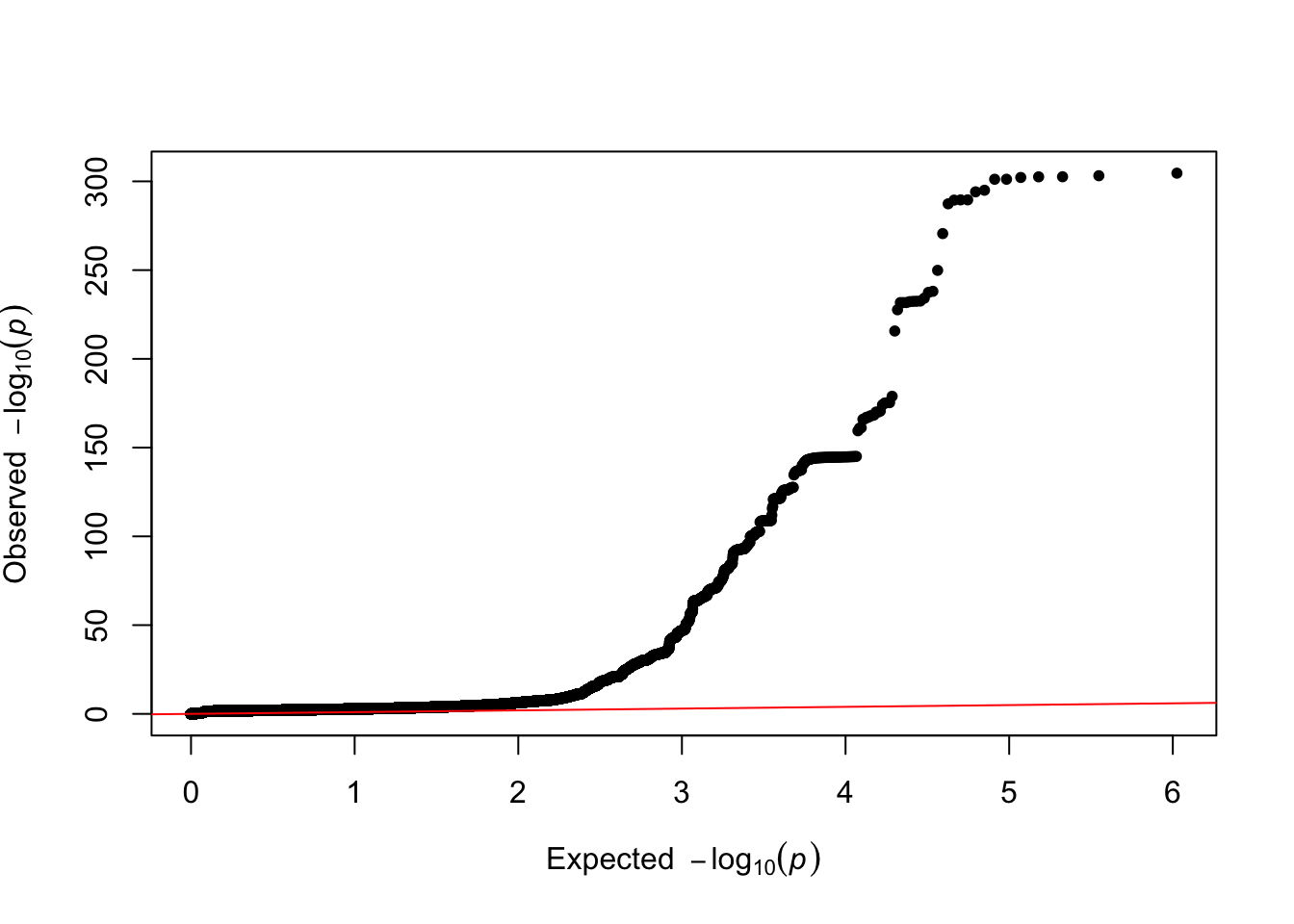
LDSC
tmp=read.table("results/stratifiedGWAS/vitD_winterGWAS.ldsc.log", skip=15, fill=T, h=F, colClasses="character", sep="_")[,1]
# Get results:
h2=strsplit(grep("Total Observed scale h2",tmp, value=T),":")[[1]][2]
lambda_gc=strsplit(grep("Lambda GC",tmp, value=T),":")[[1]][2]
chi2=strsplit(grep("Mean Chi",tmp, value=T),":")[[1]][2]
intercept=strsplit(grep("Intercept",tmp, value=T),":")[[1]][2]
ratio=strsplit(grep("Ratio",tmp, value=T),":")[[1]][2]- Total Observed scale h2: 0.0862 (0.0134)
- Lambda GC: 1.1973
- Mean Chi2: 1.2942
- Intercept: 1.0167 (0.0088)
- Ratio: 0.0569 (0.0298)